Publications
On the shape and connections of micelles: electron microscopy imaging inspiring thermodynamic modelling
Danino D, Zemb T. Curr Opin Colloid In 101607 (2022).
We illustrate in this review the dynamic interplay, in the last three decades, between progress in imaging of dynamic objects such as micelles and the ripening of predictive models of free energy depending on shape and interactions between micelles. The concept of pseudo-phases by Charles Tanford was the first to successfully explain the sphere-to-rod transition. The concept was further developed in a variety of colloidal systems by Peter Kralchevsky and coworkers. Considering the cylindrical parts, flat parts, rims, connection points, and endcaps as pseudo-phases, a large variety of morphologies observed by staining-free cutting-edge electron microscopy techniques could be considered as different morphologies resulting from Boltzmann equilibria between pseudo-phases.
We dedicate this paper to Professor Peter Kralchevsky, in appreciation of his many seminal contributions to colloid and interface science, including the enlightening works on micellar systems that had a major impact on both the basic understanding of fundamental concepts and the applications of these systems.

A Combinatorial Library of Lipid Nanoparticles for RNA Delivery to Leukocytes
Ramishetti, S., Hazan‐Halevy, I., Palakuri, R., Chatterjee, S., Naidu Gonna, S., Dammes, N., Freilich, I., Kolik Shmuel, L., Danino, D., Peer, D. Advanced Materials 32 (12), 1906128 (2020)
Lipid nanoparticles (LNPs) are the most advanced nonviral platforms for small interfering RNA (siRNA) delivery that are clinically approved. These LNPs, based on ionizable lipids, are found in the liver and are now gaining much attention in the field of RNA therapeutics. The previous generation of ionizable lipids varies in linker moieties, which greatly influences in vivo gene silencing efficiency. Here novel ionizable amino lipids based on the linker moieties such as hydrazine, hydroxylamine, and ethanolamine are designed and synthesized. These lipids are formulated into LNPs and screened for their efficiency to deliver siRNAs into leukocytes, which are among the hardest to transfect cell types. Two potent lipids based on their in vitro gene silencing efficiencies are also identified. These lipids are further evaluated for their biodistribution profile, efficient gene silencing, liver toxicity, and potential immune activation in mice. A robust gene silencing is also found in primary lymphocytes when one of these lipids is formulated into LNPs with a pan leukocyte selective targeting agent (β7 integrin). Taken together, these lipids have the potential to open new avenues in delivering RNAs into leukocytes.
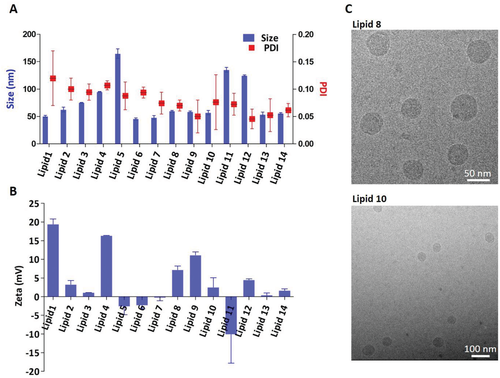
Physico‐chemical properties of LNPs made from different ionizable lipids (lipids 1–14). a) LNPs mean diameter and PDI; b) zeta potential measured by Zeta Sizer; c) cryo‐TEM analysis of lipid‐8‐ and lipid‐10‐based LNPs.
Shape and fluctuations of frustrated self-assembled nano ribbons
Zhang, M., Grossman, D., Danino, D., and Sharon, E. Nat Commun, 10(1):1-7 (2019)
Self-assembly is an important process by which nontrivial structures are formed on the sub-micron scales. Such processes are governed by chemical and physical principles that dictate how the molecular interactions affect the supramolecular geometry. Currently there is no general framework that links between molecular properties and the supramolecular morphology with its size parameters. Here we introduce a new paradigm for the description and analysis of supramolecular structures that self-assemble via short-range interactions. Analysis of molecular interactions determines inputs to the theory of incompatible elasticity, which provides analytic expressions for supramolecular shape and fluctuations. We derive quantitative predictions for specific amphiphiles that self-assembled into chiral nanoribbons. These are quantitatively confirmed experimentally, revealing unique shape evolution, unusual mechanics and statistics, proving that the assemblies are geometrically incompatible. The success in predicting equilibrium and statistics suggests the approach as a new framework for quantitative study of a large variety of self-assembled nanostructures.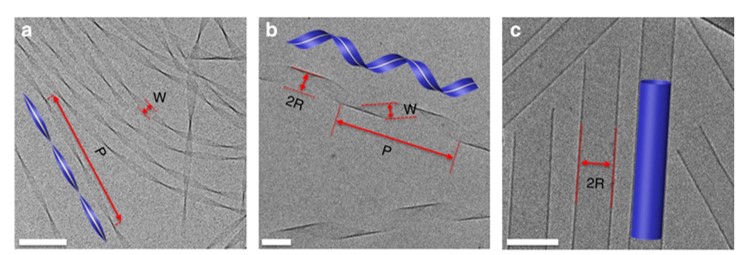
Shape evolution and characterization: Cryo-TEM images and illustrations (insets) of self-assembled N- α -lauryl-lysyl-aminolauryl-lysyl-amide (C12-β12) ribbons. a After ~24 h of assembly most ribbons are twisted, having a straight centerline, (inset, yellow dashed line) i.e. R = 0. b After 1 week, helical ribbons are abundant. Their center line is a helix with given pitch, P, and radius, R. Determination of W, P, and R from the image is demonstrated. c After 5 months most assemblies are tubes (distinguished by the dark parallel boundaries compared to the pale ends) with diameters D = 2 R ≈ 100 nm. Scale bars = 100 nm
Congratulations

Winning one of four research grants from Materna Institute on “Following Structure Formation During the Digestion of Infant Formula by Cryo-TEM”

Luba Kolik receiving the Litan Prize, with Litan family and dean Prof. Yuval Shoham of Biotechnology and Food Engineering faculty .

Inbar Elazar receiving Chairman award for excellence, with the Technion Chairman – Prof. Peretz Lavie.